The dotted line across the figure indicates the average energies for each peptide and all parts below this line are considered hydrophobic. Positively and negatively charged residues are indicated by arrows pointing upwards and downwards, respectively. capsulatus (solid line or arrows) X protein hydropathy plots were generated using the mean of 11 amino acids. sphaeroides (dotted line or arrows) and R. 5.HYDROPATHY PLOTS OF THE DERIVED X GENE PEPTIDES. At pH 7 the side chains of these amino acids carry charges-positive for arginine and lysine, negative. The amino acids are referred to as hydrophilic amino acids and include arginine, lysine, aspartic acid, and glutamic acid. Īt the surfaces of proteins are amino acid residues that interact with water.
It ranges between less than 3 and more than 12. The protein isoelectric point (pi) value is the pH at which the sum of acidic amino-add negative charges and of basic amino-add positive charges equals zero.
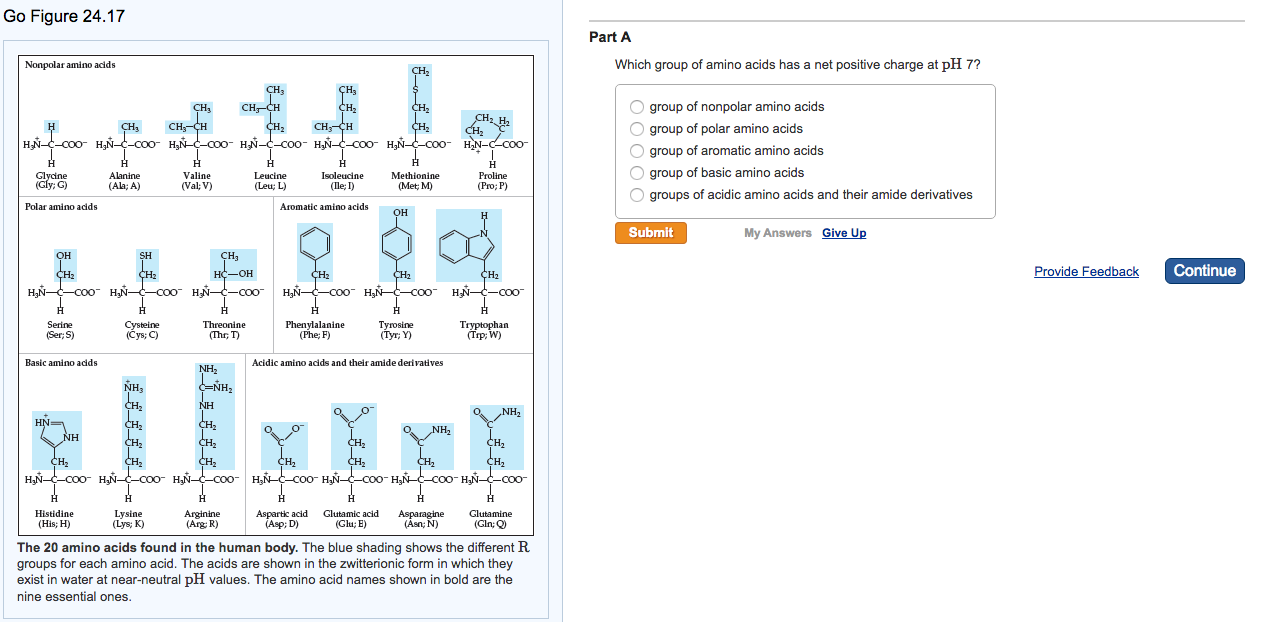
Positively charged amino acids trial#
The optimal combination of techniques is usually reached by trial and error. Protein purification techniques exploit differences in size, shape, charge, solubility, and specific binding affinity of the proteins. Mixtures of proteins will yield mixtures of peptides and ambiguous amino acid positions a unique amino acid sequence can be obtained only from a pure protein. Purification of a protein is indispensable for establishing its amino acid sequence. (Redrawn with permission from Innerarity ei al., 1984.). All the highlighted amino adds aie positively charged and occur at the apo E binding site, that is, that portion of the molecule tecognifed by an apo E receptor. The amino acid positions that have been found to be altered genetically in patients with cardiovascular diseases are highlighted and numbered. A silhouette of the apo E molecule is shown.
More often, these amino sugars are N-acylated, generally with acetyl groups.
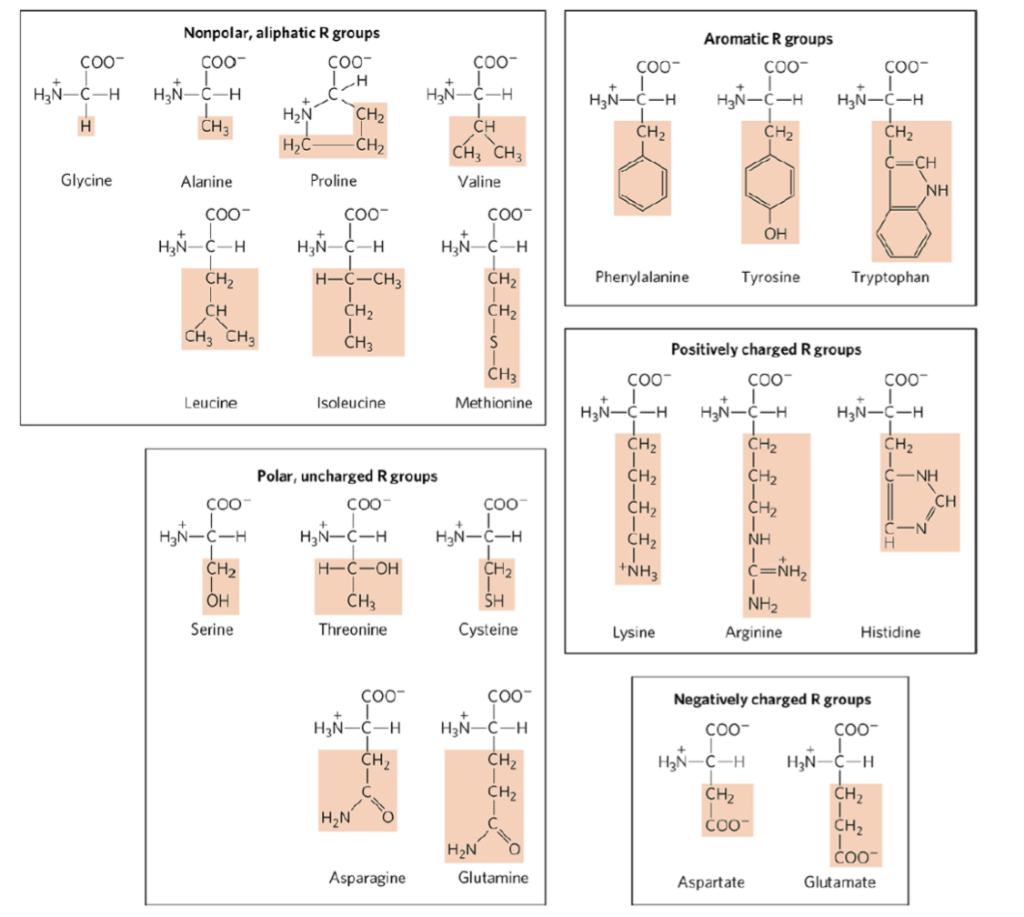
Positively charged amino acids free#
Positively charged biopolymers that contain free amino sugars are rare, but have been found (see Table VII). In many instances, these polymers possess alkali-labile O- acyl substituents, such as acetic, formic, ma-lonic, pyruvic, and succinic acids. Often, these acidic biopolymers contain other sugars, including pentoses, hexoses, and heptoses, also found in neutral polysaccharides (see Tables V and VI). Īcidic polysaccharides (see Table IV) that contain uronic acid residues are, perhaps, the most prevalent type of exocellular polysaccharide. Some cationic antimicrobial peptides also show antiviral (14) and/or anticancer activity (15) as well as wound healing properties (16). ĭespite the variety of different sources, sequences and structures, all cationic antimicrobial peptides share some common features small size (12-50 amino acids), positive net charge (+2 to +9), amphiphilic (>30% hydrophobic amino acids) and antimicrobial and/or immunomodulatory activity. Positively charged amino acids are bad substrates, while negatively charged D- amino acids are not substrates.In contrast, methionine and valine are the best substrates for RgDAAO.
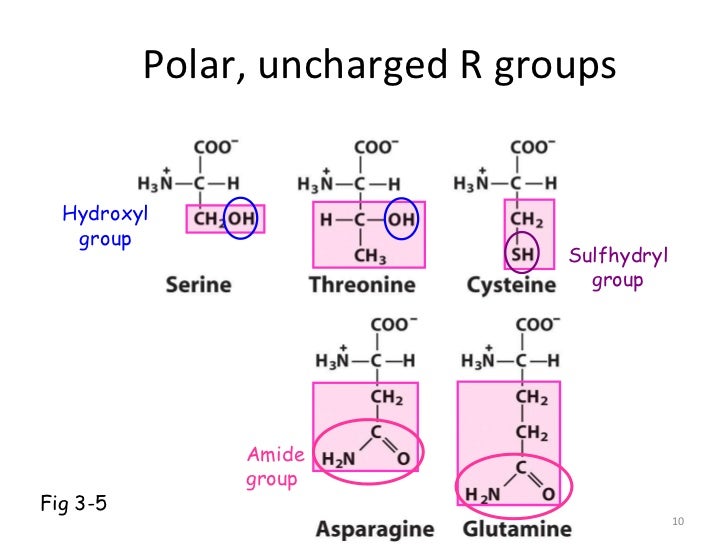
The best substrate for pkDAAO is D-proline, followed by hydrophobic and neutral amino acids. Each has different substrate specificities. More recently, yeast DAAOs from Rhodotorula gracilis (RgDAAO) and Trigonopsis variabilis (TvDAAO) have also been studied. The first DAAO studied mechanistically was from pig kidney (pkDAAO) many kinetic and mechanistic studies have been performed on this enzyme.
